FDA Investigator Barbara Janine Breithaupt
Barbara Janine Breithaupt has conducted inspections on 176 sites in 16 countries as of 17 May 2021. See below for a list of the FDA enforcement documents resulting from those inspections.
Investigator Details
Number of Inspected Sites:
176
Last Inspection Date:
17 May 2021
Investigator Role:
FDA Investigation Participant
Redica ID:
Countries of Inspections:
United States of America,
Taiwan,
Germany,
Poland,
Canada,
Netherlands,
Sweden,
China,
India,
South Africa,
New Zealand,
Switzerland,
Austria,
Australia,
Ireland,
Thailand
FDA Investigators that have inspected at least one site in common with Barbara Janine Breithaupt:
Adam R Cooke,
Adetutu M Gidado,
Adree N Anderson,
Adrienne Morgan,
Alan P Kurtzberg,
Alanna L Mussawwir Bias,
Alexander M Kay,
Alia Legaux,
Alicia M Frees,
Allen F Hall,
Allison V Bonnenburg,
Alysia C Alger,
Amanda E Lewin, PhD,
Andrace L Deyampert,
Andrea A Branche,
Andrew "Drew" Haack, PhD,
Andrew A Hoopes,
Andrew K Haack, PhD,
Andrew Le,
Andrew R Wasko,
Angela E Glenn,
Anita Narula, PhD,
Anita R Michael,
Ann L Demarco,
Ann Marie Montemurro,
Ann Marie Schofield,
Anna L Tucker,
Anna Lazar,
Annette L Diggs,
Aqualia L Nelson,
Arie C Menachem,
Ashar P Parikh,
Astrida B Mattson,
Ava M Collins,
Azza Talaat,
Babatunde Ojo,
Barbara J Holladay,
Barbara M Frazier,
Barry H Phillips,
Bo Chi, PhD,
Bonita S Chester,
Bradley Dworak, PhD,
Brandi E Williams,
Brenda W Uratani, PhD,
Brent T Hall,
Brett R Havranek,
Brian Church,
Brian M Janelsins, PhD,
Brittany D Phillips,
Brittany D Terhar,
Brittany L Carpenter,
Bruce E Taylor,
Bruce H Mccullough,
Bryan L Mcguckin,
Byungja E Marciante,
Candace S Tucker,
Carl A Anderson,
Carl A Huffman, III,
Carl J Montgomery,
Carla A Norris,
Carla J Lundi,
Carmen Y Fisher,
Carol S Sanchez,
Caryn M Mcnab,
CDR Donald Ertel,
CDR Ileana Barreto Pettit,
Chaltu Nwakijra,
Charles M Edwards,
Charles R Bonapace, PhD,
Charles R Cote, RIC,
Charles Yuanchia Kuo, PhD,
Chava Kimchi Sarfaty, PhD,
Chen Sun, PhD,
Chris A Sack,
Christian D Lynch (CDL),
Christine A Harman, PhD,
Christine M Whitby, CSO,
Christopher D Snyder,
Christopher R Czajka,
Christopher S Keating,
Clifford F Long,
Cody J Alley,
Colleen F Hoyt,
Collins M Mason,
Conner N Mann,
Connie P Rezendes,
Courtney N Long,
Ct Viswanathan, PhD,
Cynthia A Harris, MD, RN,
Cynthia J Lee, MS,
Cynthia Jim, CSO,
Cynthia M Goudeau,
Dana D Carter, SR DDS,
Dana M Klimavicz,
Dandan Wang, PhD,
Daniel J Lahar,
Daniel J Roberts,
Daniel L Aisen,
Daniel R Azar,
Darla J Christopher,
Darrah,
Darren S Brown,
David G Eng,
David J Daworski,
David L Chon,
David L Miser,
David M Beltran,
Dawn C Olenjack,
Dawn E Barkans,
Dawn L Wydner,
Deborah A Greco,
Deborah L Wade,
Deborah M Trout,
Debra Bower,
Dennis Cantellops Paite,
Diana K Krepel,
Diane T Wade, MS,
Dina K West,
Dina Kloost,
Djamila Harouaka,
Dolores E Price,
Don A Brunssen,
Don H Bark, PhD,
Donald B Mckechnie,
Donald C Obenhuber, PhD,
Donald W Jehn,
Dongping Dai, PhD,
Douglas A Campbell,
Dr. Chunchang Fang,
Dr. Gopa Biswas, PhD,
Dr. Mark J Seaton, PhD,
Dr. Ralph M Bernstein, PhD,
Dr. Sriram Subramaniam, PhD,
Dr. Zhou Chen (nmi), MD PhD,
Dylan D Yao, MD, PhD,
Edmund F Mrak, Jr,
Edward E Lockwood (EEL),
Edwin Martinez,
Edwin Melendez,
Eileen A Liu,
Elizabeth D Connell,
Emest F Bizjak,
Eric C Nielsen,
Eric L Dong, BS,
Eric M Mueller, PharmD,
Eric S Weilage,
Eric T Huebler,
Erika V Butler,
Erin L Mcfiren,
Erin M Mcdowell,
Esaw,
Esteban Beltran,
Esther A Ofori,
Farhana Khan,
Gary C Pecic, Jr,
Gene D Arcy,
George G Calafactor,
Gerald B Seaborn, Jr,
Gerard Pde Leon,
Ginger M Sykes,
Gloria J Baca, MS,
Grace P Santos,
Gretchen Lf Trendel,
Gwyn G Dickinson,
Hala L Selby,
Heath W Cartwright,
Heika R Bounds,
Heika R Tait,
Hugh M Mcclure, II,
Iris C Macinnes,
Jacek Cieslak, PhD,
Jacqueline A O'shaughnessy, PhD,
Jacqueline Mdiaz Albertini,
James A Lane,
James D Bridges,
James D Planchon,
James I Giefer,
James J Houlihan,
James L Dunnie, Jr,
James S Stuart, BS,
Jane M Kreis,
Janesa A Licorish,
Janete A Eaker,
Janete A Oliveira,
Janete F Guardia,
Jeffery A Hangartner,
Jeffrey P Raimondi,
Jen Sui,
Jennifer C Adams,
Jennifer L Bridgewater,
Jennifer S Ness,
Joan A Loreng,
Joan Johnson,
Joan M Cantellops Figueroa,
Joanne E King,
Joey C West,
Joey V Quitania,
John A Iwen,
John M Gusto,
Johnson,
Jonathan W Chapman,
Jorge L Guadalupe,
Jose A Lopez,
Jose Acruz Gonzalez,
José E Meléndez,
Jose M Cayuela,
Jose Martinez, Jr,
Joselin P Baray Alvarado,
Joseph R Lambert,
Joshua P Wireman,
Juanita P Versace,
Julian C Hanson,
Julie D Bringger,
Junho Pak,
Jyoti B Patel, PhD,
Kalavati Suvarna, PhD,
Kamara Mo,
Karen C Daugherty,
Karen E D'orazio,
Karen M Montgomery (KMM),
Kari M Johansen,
Katherine M Taylor,
Katherine Szestypalow,
Kathleen B Swat,
Kathleen J Close,
Kathleen R Jones, PhD,
Kathryn A Krentz,
Kathryn E King,
Kejun Cheng,
Kellia N Hicks,
Kellie L Thommes, RN,
Kenneth Nieves,
Kenneth O Gee, PhD,
Kevin A Beavers,
Kevin A Gonzalez,
Kevin D Kallander,
Kevin E Klommhaus,
Kevin P Foley,
Kham Phommachanh,
Kimberley A Hoefen,
Krishna Ghosh, PhD,
Kristen D Evans,
Kristi B Panzer,
Kristin M Abaonza,
Kristopher J Stanya, PhD,
Kurt A Brorson, PhD,
Lakisha M Williams,
Larry K Austin,
Laurel A Beer,
Lauren E Swantko,
Laurie A Haxel,
Laverne Puckett,
Lawrence J Stringer,
Lawrence Y Lee, PhD,
LCDR Debra Emerson,
LCDR Ismael Olvera, IV,
Lesley A Swanson,
Li Hongpaul Yeh, PhD,
Linan Ha, PhD,
Linda K Cline,
Linda R Kuchenthal,
Linda S Leja,
Linda Thai,
Lindsey K Giles Austin,
Linhart,
Lionell L Thomas,
Lisa K Capron,
Lisa L Flores,
Lisa P Oakes,
Lisa T Michel,
Lloyd M Luapula,
Lori A Gioia,
Lori J Silverstein,
Luis Mburgos Medero,
Lynda L Perry, PhD,
Lynette P Salisbury,
Mai X Huynh,
Makini Cobourne Duval, PhD,
Marci M Deleon,
Margaret M Annes,
Maria A Ruttell,
Maria Gutierrez Lugo, PhD,
Maria Joselopez Barragan, PhD,
Maria Pkelly Doggett, MBA,
Maridalia Torres Irizarry,
Marie B Buen Bigornia,
Marie F Morin,
Marijo B Kambere, PhD,
Marilyn M White,
Marion W Nadeau,
Marisol Faberlle,
Mariza M Jafary,
Marjorie A Shapiro, PhD,
Mark W Babbitt,
Marlene G Swider,
Marquessa M Rowe,
Martin K Yau, PhD,
Marvin D Jones,
Mary E Farbman, PhD,
Mary K Concannon,
Mathur,
Matthew B Casale,
Maxine H Wong,
Megan A Haggerty,
Meisha R Sampson,
Meisha Waters,
Melinda L Rice,
Melissa J Garcia,
Michael A Charles,
Michael A Feingold,
Michael E Maselli,
Michael F Skelly, PhD,
Michael J Verdi,
Michael P Sheehan,
Michael R Goga,
Michael S Araneta,
Michael Shanks, MS,
Michele L Obert,
Michele Perry Williams,
Michelle Yclark Stuart,
Miguel A Martinez Perez,
Mihaly S Ligmond,
Mikel T Wright,
Mikhail V Ovanesov, PhD,
Mindy M Chou,
Minerva Rogers,
Mohsen Rajabi Abhari, FDA,
Monica Cburgos Garcia,
Mra B Kamberem,
Mra Mcculloughj,
Muralidhara B Gavini, PhD,
Nancy A Bellamy,
Nancy A Saxenian Emmons,
Nancy E Byerly,
Nancy G Schmidt,
Naseem S Jouhari,
Nataniel Phillips Sylvain, PhD,
Nathan R Moon,
Nebil A Oumer,
Ngoc B Kelsch,
Nia K Walker,
Nicholas L Paulin,
Nicola M Fenty Stewart,
Nilufer M Tampal, PhD,
Norman K Starks,
Omotunde O Osunsanmi,
Otto D Vitillo,
Pal S Mayasandra,
Parul M Patel,
Patrick C Klotzbuecher,
Patrick J Lynch,
Patrick L Wisor,
Patsy J Domingo,
Paul A Bonneau,
Paul L Bellamy,
Paul L Figarole, Jr,
Paul P Geraci,
Paula A Trost,
Paula J Bretz,
Peter Abel,
Peter C Chow,
Peter E Gruman,
Philip F Istafanos, DMV, MS,
Philip J Snoy,
Ponzoj,
Prabhu P Raju,
Qin Xu,
Rabia Ballica, PhD,
Rachel C Harrington,
Ralph Jerndal,
Randy L Self,
Rashonda N Rucker,
Raymond T Anderson,
Raymond T Oji,
Rebecca E Dombrowski,
Rebecca Rodriguez,
Reyes Candau Chacon, PhD,
Richard E Needham,
Richard Heath Coats,
Richard Jay Tucker, RS,
Richard K Vogel,
Richard L Friedman,
Richard Ledwidge (nmi), PhD,
Riley C Myers, PhD,
Rita K Kabaso,
Robert D Tollefsen,
Robert J Waldorf,
Robert L Hummel,
Rochelle K Kimmel,
Rodney D Combs,
Rodney G Raiford,
Roger F Zabinski,
Ron R Pearson,
Ronald G Crawford,
Rose Ashley,
Rose Xu,
Russell J Glapion,
Ryan S Martin,
S Lori Brown, PhD MPH,
Saied A Asbagh,
Saleem A Akhtar,
Sangeeta M Khurana, PhD,
Sangeeta M Rataul,
Santos E Camara,
Sarah E Mcmullen,
Sarah E Rhoades,
Sarah Forney,
Sarah Ibrahim,
Satheesh Thomas,
Scot M Stodola,
Scott A Jepson,
Scott B Laufenberg,
Scott Platt,
Scott R Nichols, PhD,
Scott T Ballard,
Sean R Marcsisin,
Seneca D Toms,
Sergio Chavez (NMI),
Shanna R Ausfahl,
Shanna R Haden,
Sharon K Thoma, PharmD,
Shaun M Olson,
Shawn B Johnson,
Sherri N Rohlf, MD,
Sherry L Secrist, BLT DO,
Shirley H Isbill,
Shirley J Berryman,
Shuen G Chai,
Simone E Pitts,
Sneha S Patel,
Soyinka S Eneng,
Sripal R Mada, PhD,
Stephanie L Shapley,
Stephen D Eich,
Steven A Gonzales,
Steven C Clifford,
Steven C Madzo,
Steven D Kehoe,
Steven Fong, MS, PhD,
Steven M Weinman,
Steven P Allen,
Steven P Donald,
Steven W Brown,
Stroman,
Sunitha K Rajaram, PhD,
Susan F Laska, MS,
Susan M Jackson,
Susan T Hadman,
Susanna E Ford,
Tamara N Champion,
Tara G Bizjak,
Tara L Greene,
Tara L King,
Tawny L Colling,
Teegan Dellibovi Ragheb, PhD,
Terri L Dodds,
Theressa B Smith,
Thomas J Arista,
Thora W Whitmore,
Thuy T Nguyen, LCDR,
Tiffani D Wilson,
Tiki J Dixon,
Tina S Roecklein,
Torrance J Slayton,
Torrey M Ward,
Toyin B Oladimeji,
Tracy K Li,
Tristian E Strait,
Truong Xuan Nguyen (Andy),
Uttaniti Limchumroon (Tom),
V Teres Speer,
Vaishali J Patel,
Vanessa Y Gelsey, PhD,
Vanessa Y Jacobs,
Vickie J Kanion,
Victoria L Palmer,
Vilmary Negron Rodriguez,
Walden H Lee,
Walsworth,
Wanda B Coats,
Wayne D Mcgrath,
Wayne E Seifert,
Wayne T Smith,
Wendy G Tan, PhD,
Xianghong Jing (Emily), PhD,
Xikui Chen (nmi), PhD,
Xiomara Copeland,
Yideng Liang,
Yong Hu,
Yuan Chia Kuo,
Yumi J Hiramine,
Zachary A Bogorad,
Zhong Li, PhD,
Zhongren Wu
Barbara Janine Breithaupt's Documents
Experience Redica Systems’ NEW investigator profiles and dashboards
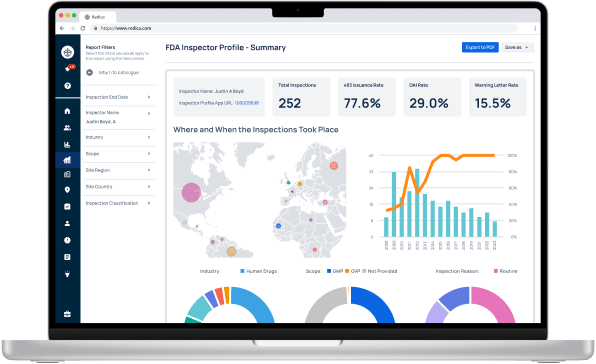
Redica Systems customers get access to over 1,400 FDA Investigator profiles, containing powerful analysis like:
- Total inspections conducted
- 483 rate
- OAI rate
- Warning Letter rate
- Inspections by Industry, Scope, and Inspection Reason
- And much more
Investigator Profiles are not sold as on-demand documents. If you’d like to see one, fill out the form below to connect with our Sales Team and get it in your inbox as little as 24 hours.
Talk to sales to access our investigator profiles
Loading...
Investigator Profiles Are Just the Tip of the Iceberg
Becoming a Redica Systems customer not only provides you with unlimited access to profiles similar to the one above for every current and former FDA Investigator, but also grants you access to:
Inspection Intelligence
- Pre-Approval Inspection (PAI) trends
- The ability to filter nearly any FDA enforcement action by GxP labels like GMP, GCP, etc.
- Latest 483s: see what the trends are across all FDA Investigators
- CFR Heatmap: Citations aggregated by Subpart by Year
Vendor Intelligence
- Full inspection histories for your vendors down to the specific site level
- All of the functionality mentioned above but isolated to just your vendors
Regulatory Intelligence
- Zero in on the trends for a specific agency/regulator (ex. FDA, MHRA, EMA, Health Canada)
- All the latest "Signals" from agencies around the world, segmented by Country, Type (ex. Guidance, Decree...), Category (ex. Rules/Regulations/Guidance, News, Reports...), Theme (ex. Cell and Gene Therapy, AI...), and more